Cryptosporidiosis: A Significant Threat to One Health
Thank you for reading this post, don't forget to subscribe!Munwar Ali 1, Ali Hassan Nawaz2, Kun Li 1
1 College of Veterinary Medicine, Nanjing Agricultural University, Nanjing 210095, PR China; drmunwarali06@gmail.com, lk3005@njau.edu.cn
2 College of Animal Science and Technology, Nanjing Agricultural University, Nanjing 210095, PR China ah93163@gmail.com
Abstract
Cryptosporidium spp. is a protozoan parasite of critical One Health importance, posing a substantial threat to global public health and causing significant economic losses in the livestock industry. This extension article synthesizes current knowledge to comprehensively assess the cryptosporidiosis landscape. We elucidate its complex life cycle and high prevalence rates in human and animal populations, underscoring its profound zoonotic potential. The analysis highlights the primary transmission routes, particularly through water and food, by examining major global outbreaks to demonstrate the scale of contamination risk. The significant production losses associated with animal infections are also quantified. Crucially, this article consolidates integrated preventive and control strategies, research gaps, and future perspectives.
Introduction
Cryptosporidium, a genus of protozoan parasites, is responsible for cryptosporidiosis. It was first discovered in the early 20th century [1] and later attracted greater attention due to its increasing prevalence, wide host range, zoonotic potential, and its unique characteristics, which proved lethal in immunocompromised individuals [2]. It ranked fifth among 24 foodborne parasites worldwide [3]. Contaminated drinking and recreational water [4,5], consumption of vegetables [8], raw milk, and apple cider [5] lead to outbreaks. During 2010 to 2020, in 96.5% of Cryptosporidium-food-related pathologies, C. parvum was detected as an etiological agent [9].
The parasite can infect fish, amphibians, reptiles, birds, and mammals [10], resulting in huge economic losses to the veterinary sector [11] and zoonotic transmission. The 1993 Milwaukee waterborne outbreak affected 400,000 individuals with 69 deaths [12,13]. Between 2011-2016, 63% of Cryptosporidium-related waterborne outbreaks led to 4.2 million disability-adjusted life years (DALYs) [14]. According to the Global Enteric Multicenter Study (GEMS), cryptosporidiosis ranked as the second leading cause of diarrhea in infants, responsible for approximately 202,000 deaths annually [15]. Overall, cryptosporidiosis is an ever-increasing challenge to the food and water industry, negatively influencing both public health and livestock sectors [3,16].
Potential Risk Factors Fostering Cryptosporidium Spread
The spread of Cryptosporidium infection is linked to contact with contaminated animals, age, gender, poverty, illiteracy, sexual practices, population density, seasonal variations, swimming, immune status, occupational activities, natural disasters, and political disputes [16,17]. Seasonal variations rely on temperature, parasitic species, and water source [18]. Spring peaks may indicate livestock activities like calving, while late-summer surges correlate with recreational water use [19,20].
Infectious Life Cycle of Cryptosporidium spp.
The life cycle culminates in fecal release of contagious oocysts [24]. Upon ingestion, excystation releases sporozoites that invade intestinal epithelial cells, undergoing asexual (merogony) and sexual (gametogony) reproduction [25]. This produces thick-walled oocysts (shed into the environment) and thin-walled oocysts (causing autoinfection) [26]. The new generation can emerge in 12-14 hours [27]. Infection causes villus atrophy, inflammation, and secretory diarrhea [28,29]. In immunocompromised patients, infection can become chronic and extra-intestinal (Figure 1) [30].
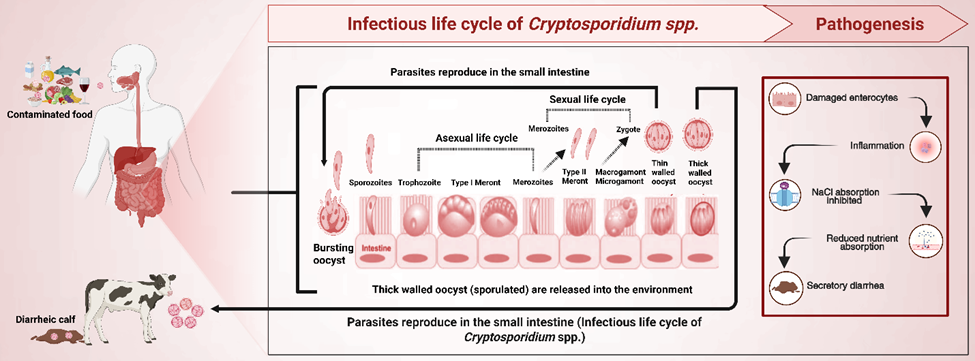
Figure 1. Infectious life cycle and pathogenesis of Cryptosporidium species
Cryptosporidium oocysts, after ingestion, complete their asexual and sexual life cycle in the epithelial cells. Upon completion of the infectious life cycle, thin-walled oocysts usually cause autoinfection within the same host, while thick-walled oocysts are released in the environment and infect new susceptible hosts (e.g., calves).
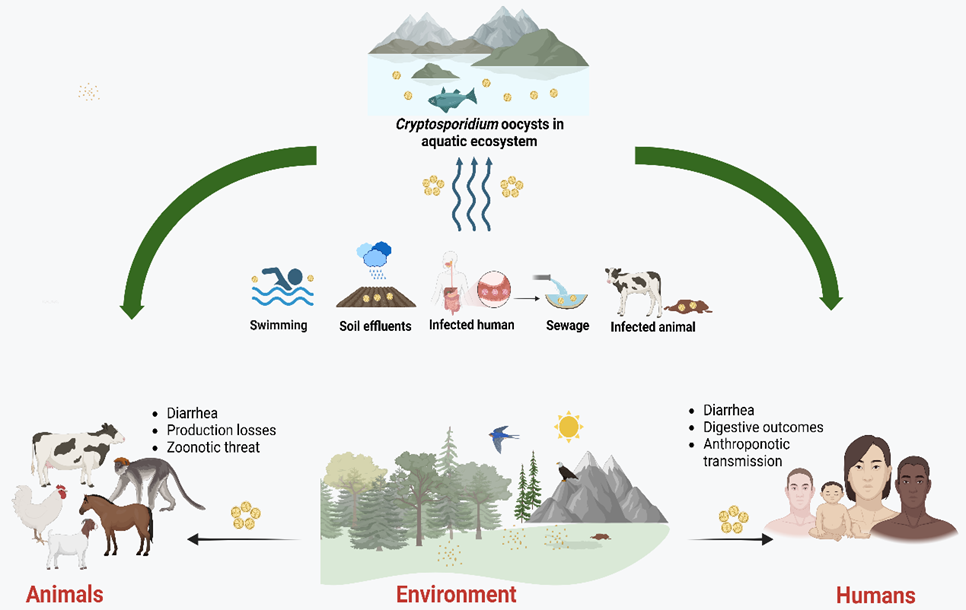
Zoonotic Transmission: Water and Foodborne
Humans are sensitive to diverse Cryptosporidium spp., particularly C. parvum and C. hominis [31,32]. C. parvum has an extremely wide host range and significant zoonotic potential due to livestock interactions [9,33]. C. andersoni has been found in humans in China [34,35], and C. bovis has been detected in humans with livestock contact [36,37]. C. suis has been detected in HIV patients in several countries [38,39]. Contaminated water is a primary vehicle for cryptosporidiosis (Figure 1); oocysts are highly resistant to chlorine, allowing them to bypass conventional water treatment [5,40].
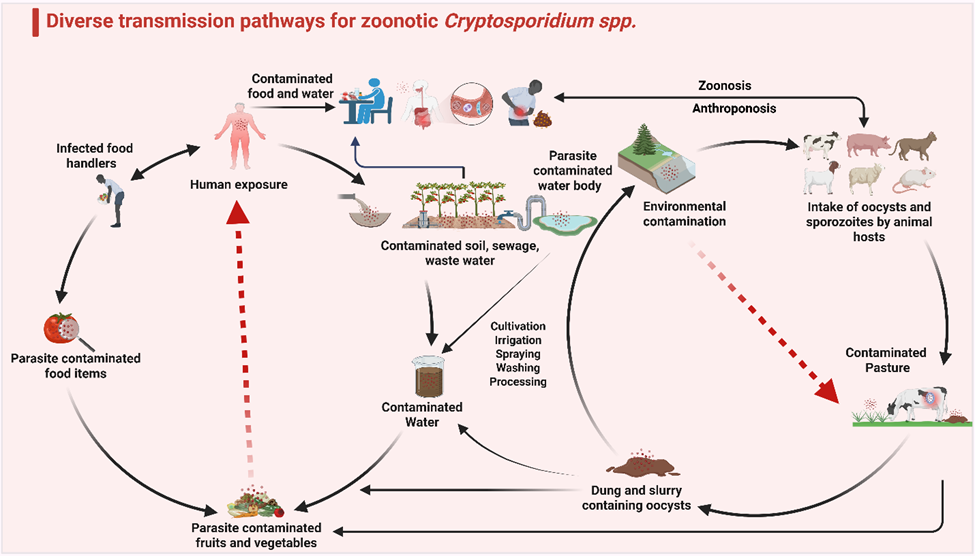
Figure 2. Diverse pathways for the spread of Cryptosporidium oocysts
This figure shows different transmission routes of Cryptosporidium oocysts between humans and animals involving different agricultural practices. By breaking transmission routes, the outbreaks can be prevented.
Cryptosporidium and Public Health
Cryptosporidium is classified as the 5th most significant foodborne parasitic infection globally [3]. Food exposure may occur during manufacturing, with oocysts harbored on fresh produce [41]. Swedish foodborne epidemics have been linked to contaminated arugula, parsley, and spinach juice [42,43]. Agriculture contributes to water contamination through farm-based effluents [44,45]. The use of contaminated water for irrigation and animal manure as fertilizer facilitates the spread [46]. Wildlife may also transmit C. parvum to the environment [47].
Cryptosporidium and Livestock
Prevalence of Cryptosporidium spp. is high: 27.8–60.4% in pigs, 18% in calves, and 28.5% in cattle [48,49]. In calves, species prevalence is age-dependent; C. parvum infects young calves, while C. bovis is prominent in older calves [50]. C. hominis has been detected in cattle, sheep, goats, and other animals, highlighting potential animal reservoirs for human infections [33,51]. Molecular studies have identified various species in wildlife, including C. scrofarum in wild boars and C. ubiquitum in hedgehogs [52,53], emphasizing the complex transmission cycles.
Preventive and Control Measures
Due to a lack of effective medications or vaccines, precautionary steps are essential. These include hand hygiene, boiling water, avoiding untreated water and raw food, and disinfecting contaminated surfaces. For animals, rotational grazing, colostrum management, and isolation of sick animals are recommended. Waste treatment methods like composting and anaerobic digestion reduce oocyst burden. Adopting a circular economy approach for agricultural waste is encouraged. Epidemiological investigations and One Health approaches are critical for control.
Research gaps and Future Perspectives
While the immunofluorescence antibody test (IFAT) is reliable, cost-effective molecular methods like PCR targeting the gp60 gene are needed for routine surveillance. Next-generation sequencing (NGS) can improve diagnostic accuracy. Research on gp60 protein host interactions, gut microbiota changes during infection, and genetic manipulation using CRISPR/Cas9 may reveal new therapeutic targets. MicroRNA regulation appears significant in host defense. Systematic studies on anti-Cryptosporidium mechanisms in different herds are needed. Better molecular understanding of transmission dynamics will aid in developing precisely adjusted control plans.
Conclusion
A variety of Cryptosporidium species and genotypes, with a broad host range, pose a major threat to food and water safety. Their zoonotic potential creates an increasing burden on public health and veterinary sectors. Frequent outbreaks worldwide call for urgent control measures. Current therapeutics are insufficient; development of an effective vaccine, improved control strategies, and potent drugs are essential to reduce cases and eliminate outbreaks.
References
- Leitch, G.J.; He, Q. Cryptosporidiosis-an Overview. J. Biomed. Res. 2012, 25, 1–16.
- Gerace, E.; Presti, V.D.M. Lo; Biondo, C. Cryptosporidium Infection: Epidemiology, Pathogenesis, and Differential Diagnosis. Eur. J. Microbiol. Immunol. 2019, 9, 119–123.
- Organization, W.H. Multicriteria-Based Ranking for Risk Management of Food-Borne Parasites: Report of a Joint FAO; FAO, World Health Organization, 2014.
- Moreira, N.A.; Bondelind, M. Safe Drinking Water and Waterborne Outbreaks. J. Water Health 2017, 15, 83–96.
- Gharpure, R.; Perez, A.; Miller, A.D.; Wikswo, M.E.; Silver, R.; Hlavsa, M.C. Cryptosporidiosis Outbreaks—United States, 2009–2017. Am. J. Transplant. 2019, 19, 2650–2654.
- Taha, S.; Elmalik, K.; Bangoura, B.; Lendner, M.; Mossaad, E.; Daugschies, A. Molecular Characterization of Bovine Cryptosporidium Isolated from Diarrheic Calves in the Sudan. Parasitol. Res. 2017, 116, 2971–2979.
- Ali, M.; Xu, C.; Nawaz, S.; Ahmed, A.E.; Hina, Q.; Li, K. Anti-Cryptosporidial Drug-Discovery Challenges and Existing Therapeutic Avenues: A “One-Health” Concern. Life 2024, 14, 80.
- Sim, S.; Won, J.; Kim, J.-W.; Kim, K.; Park, W.-Y.; Yu, J.-R. Simultaneous Molecular Detection of Cryptosporidium and Cyclospora from Raw Vegetables in Korea. Korean J. Parasitol. 2017, 55, 137.
- Zahedi, A.; Ryan, U. Cryptosporidium–an Update with an Emphasis on Foodborne and Waterborne Transmission. Res. Vet. Sci. 2020, 132, 500–512.
- Holubová, N.; Zikmundová, V.; Limpouchová, Z.; Sak, B.; Konečný, R.; Hlásková, L.; Rajský, D.; Kopacz, Z.; McEvoy, J.; Kváč, M. Cryptosporidium proventriculi sp. (Apicomplexa: Cryptosporidiidae) in Psittaciformes Birds. Eur. J. Protistol. 2019, 69, 70–87.
- Castro-Hermida, J.A.; García-Presedo, I.; González-Warleta, M.; Mezo, M. Cryptosporidium and Giardia Detection in Water Bodies of Galicia, Spain. Water Res. 2010, 44, 5887–5896.
- Lefebvre, M.; Razakandrainibe, R.; Villena, I.; Favennec, L.; Costa, D. Cryptosporidium-Biofilm Interactions: A Review. Appl. Environ. Microbiol. 2021, 87, e02483-20.
- Mac Kenzie, W.R.; Schell, W.L.; Blair, K.A.; Addiss, D.G.; Peterson, D.E.; Hoxie, N.J.; Kazmierczak, J.J.; Davis, J.P. Massive Outbreak of Waterborne Cryptosporidium Infection in Milwaukee, Wisconsin: Recurrence of Illness and Risk of Secondary Transmission. Clin. Infect. Dis. 1995, 21, 57–62.
- Chique, C.; Hynds, P.D.; Andrade, L.; Burke, L.; Morris, D.; Ryan, M.P.; O’Dwyer, J. Cryptosporidium spp. in Groundwater Supplies Intended for Human Consumption–A Descriptive Review of Global Prevalence, Risk Factors and Knowledge Gaps. Water Res. 2020, 176, 115726.
- Ayed, L. Ben; Ahmed, S.A.A.; Boughattas, S.; Karanis, P. Waterborne Cryptosporidium species and Giardia Duodenalis in Resources of MENA: A Systematic Review and Meta-Analysis. J. Water Health 2024, 22, 1491–1515.
- Aldeyarbi, H.M.; Abu El-Ezz, N.M.T.; Karanis, P. Cryptosporidium and Cryptosporidiosis: The African Perspective. Environ. Sci. Pollut. Res. 2016, 23, 13811–13821.
- CDC. Parasites-Cryptosporidium (Also Known as “Crypto).
- Koompapong, K.; Sukthana, Y. Seasonal Variation and Potential Sources of Cryptosporidium Contamination in Surface Waters of Chao Phraya River and Bang Pu Nature Reserve Pier, Thailand. Southeast Asian J. Trop. Med. Public Heal. 2012, 43, 832–840.
- Lal, A.; Hales, S.; French, N.; Baker, M.G. Seasonality in Human Zoonotic Enteric Diseases: A Systematic Review. PLoS One 2012, 7, e31883.
- Ikiroma, I.A.; Pollock, K.G. Influence of Weather and Climate on Cryptosporidiosis—A Review. Zoonoses Public Health 2021, 68, 285–298.
- Ryan, U.; Zahedi, A.; Feng, Y.; Xiao, L. An Update on Zoonotic Cryptosporidium species and Genotypes in Humans. Animals 2021, 11, 3307.
- Li, X.; Zhang, X.; Jian, Y.; Wang, G.; Ma, L.; Schou, C.; Karanis, P. Detection of Cryptosporidium Oocysts and Giardia Cysts in Vegetables from Street Markets from the Qinghai Tibetan Plateau Area in China. Parasitol. Res. 2020, 119, 1847–1855.
- Ahmed, S.A.; Karanis, P. Cryptosporidium and Cryptosporidiosis: The Perspective from the Gulf Countries. Int. J. Environ. Res. Public Health 2020, 17, 6824.
- Gunasekera, S.; Zahedi, A.; O’Dea, M.; King, B.; Monis, P.; Thierry, B.; M. Carr, J.; Ryan, U. Organoids and Bioengineered Intestinal Models: Potential Solutions to the Cryptosporidium Culturing Dilemma. Microorganisms 2020, 8, 715.
- Borowski, H.; Clode, P.L.; Thompson, R.C.A. Active Invasion and/or Encapsulation? A Reappraisal of Host-Cell Parasitism by Cryptosporidium. Trends Parasitol. 2008, 24, 509–516.
- Fayer, R.; Santin, M.; Trout, J.M. Cryptosporidium in Cattle: From Observing to Understanding. 2009.
- Carmena, D. Waterborne Transmission of Cryptosporidium and Giardia: Detection, Surveillance and Implications for Public Health. Curr. Res. Technol. Educ. Top. Appl. Microbiol. Microb. Biotechnol. 2010, 20, 3–4.
- Chen, X.-M.; Keithly, J.S.; Paya, C. V; LaRusso, N.F. Cryptosporidiosis. N. Engl. J. Med. 2002, 346, 1723–1731.
- Farthing, M.J.G. Clinical Aspects of Human Cryptosporidiosis. Cryptosporidiosis and microsporidiosis 2000, 6, 50–74.
- Chalmers, R.M.; Davies, A.P. Minireview: Clinical Cryptosporidiosis. Exp. Parasitol. 2010, 124, 138–146.
- Ryan, U.; Zahedi, A.; Paparini, A. Cryptosporidium in Humans and Animals—a One Health Approach to Prophylaxis. Parasite Immunol. 2016, 38, 535–547.
- Xiao, L. Molecular Epidemiology of Cryptosporidiosis: An Update. Exp. Parasitol. 2010, 124, 80–89.
- Santin, M. Cryptosporidium and Giardia in Ruminants. Vet. Clin. North Am. Food Anim. Pract. 2020, 36, 223–238.
- Jiang, Y.; Ren, J.; Yuan, Z.; Liu, A.; Zhao, H.; Liu, H.; Chu, L.; Pan, W.; Cao, J.; Lin, Y. Cryptosporidium andersoni as a Novel Predominant Cryptosporidium species in Outpatients with Diarrhea in Jiangsu Province, China. BMC Infect. Dis. 2014, 14, 1–6.
- Liu, H.; Shen, Y.; Yin, J.; Yuan, Z.; Jiang, Y.; Xu, Y.; Pan, W.; Hu, Y.; Cao, J. Prevalence and Genetic Characterization of Cryptosporidium, Enterocytozoon, Giardia and Cyclospora in Diarrheal Outpatients in China. BMC Infect. Dis. 2014, 14, 1–6.
- Khan, S.M.; Debnath, C.; Pramanik, A.K.; Xiao, L.; Nozaki, T.; Ganguly, S. Molecular Characterization and Assessment of Zoonotic Transmission of Cryptosporidium from Dairy Cattle in West Bengal, India. Vet. Parasitol. 2010, 171, 41–47.
- Ng, J.S.Y.; Eastwood, K.; Walker, B.; Durrheim, D.N.; Massey, P.D.; Porigneaux, P.; Kemp, R.; McKinnon, B.; Laurie, K.; Miller, D. Evidence of Cryptosporidium Transmission between Cattle and Humans in Northern New South Wales. Exp. Parasitol. 2012, 130, 437–441.
- Cama, V.A.; Ross, J.M.; Crawford, S.; Kawai, V.; Chavez-Valdez, R.; Vargas, D.; Vivar, A.; Ticona, E.; Navincopa, M.; Williamson, J. Differences in Clinical Manifestations among Cryptosporidium species and Subtypes in HIV-Infected Persons. J. Infect. Dis. 2007, 196, 684–691.
- Moore, C.E.; Elwin, K.; Seng, C.; Mao, S.; Suy, K.; Kumar, V.; Nader, J.; Bousfield, R.; Perera, S.; Bailey, J.W. Molecular Characterization of Cryptosporidium species and Giardia duodenalis from Symptomatic Cambodian Children. PLoS Negl. Trop. Dis. 2016, 10, e0004822.
- Rosenthal, V.D.; Maki, D.G.; Mehta, Y.; Leblebicioglu, H.; Memish, Z.A.; Al-Mousa, H.H.; Balkhy, H.; Hu, B.; Alvarez-Moreno, C.; Medeiros, E.A. International Nosocomial Infection Control Consortium (INICC) Report, Data Summary of 43 Countries for 2007-2012. Device-Associated Module. Am. J. Infect. Control 2014, 42, 942–956.
- Ryan, U.; Hijjawi, N.; Xiao, L. Foodborne Cryptosporidiosis. Int. J. Parasitol. 2018, 48, 1–12.
- Insulander, M.; Silverlås, C.; Lebbad, M.; Karlsson, L.; Mattsson, J.G.; Svenungsson, B. Molecular Epidemiology and Clinical Manifestations of Human Cryptosporidiosis in Sweden. Epidemiol. Infect. 2013, 141, 1009–1020.
- Insulander, M.; De Jong, B.; Svenungsson, B. A Food-Borne Outbreak of Cryptosporidiosis among Guests and Staff at a Hotel Restaurant in Stockholm County, Sweden, September 2008. Eurosurveillance 2008, 13, 19071.
- Mateo-Sagasta, J.; Zadeh, S.M.; Turral, H.; Burke, J. Water Pollution from Agriculture: A Global Review. Executive Summary. 2017.
- Wato, T.; Amare, M.; Bonga, E.; Demand, B.B.O.; Coalition, B.B.R. The Agricultural Water Pollution and Its Minimization Strategies—A Review. J. Resour. Dev. Manag 2020, 64, 10–22.
- Iqbal, A.; Labib, M.; Muharemagic, D.; Sattar, S.; Dixon, B.R.; Berezovski, M. V Detection of Cryptosporidium parvum Oocysts on Fresh Produce Using DNA Aptamers. PLoS One 2015, 10, e0137455.
- García-Presedo, I.; Pedraza-Díaz, S.; González-Warleta, M.; Mezo, M.; Gómez-Bautista, M.; Ortega-Mora, L.M.; Castro-Hermida, J.A. Presence of Cryptosporidium scrofarum, C. suis and C. parvum Subtypes IIaA16G2R1 and IIaA13G1R1 in Eurasian Wild Boars (Sus Scrofa). Vet. Parasitol. 2013, 196, 497–502.
- Gattan, H.S.; Alshammari, A.; Marzok, M.; Salem, M.; AL-Jabr, O.A.; Selim, A. Prevalence of Cryptosporidium Infection and Associated Risk Factors in Calves in Egypt. Sci. Rep. 2023, 13, 17755.
- Hatam-Nahavandi, K.; Ahmadpour, E.; Carmena, D.; Spotin, A.; Bangoura, B.; Xiao, L. Cryptosporidium Infections in Terrestrial Ungulates with Focus on Livestock: A Systematic Review and Meta-Analysis. Parasit. Vectors 2019, 12, 453.
- Santín, M.; Trout, J.M.; Fayer, R. A Longitudinal Study of Cryptosporidiosis in Dairy Cattle from Birth to 2 Years of Age. Vet. Parasitol. 2008, 155, 15–23.
- Widmer, G.; Köster, P.C.; Carmena, D. Cryptosporidium hominis Infections in Non-Human Animal Species: Revisiting the Concept of Host Specificity. Int. J. Parasitol. 2020, 50, 253–262.
- Li, W.; Deng, L.; Wu, K.; Huang, X.; Song, Y.; Su, H.; Hu, Y.; Fu, H.; Zhong, Z.; Peng, G. Presence of Zoonotic Cryptosporidium scrofarum, Giardia duodenalis Assemblage A and Enterocytozoon bieneusi Genotypes in Captive Eurasian Wild Boars (Sus Scrofa) in China: Potential for Zoonotic Transmission. Parasit. Vectors 2017, 10, 1–8.
- Qi, M.; Luo, N.; Wang, H.; Yu, F.; Wang, R.; Huang, J.; Zhang, L. Zoonotic Cryptosporidium spp. and Enterocytozoon bieneusi in Pet Chinchillas (Chinchilla Lanigera) in China. Parasitol. Int. 2015, 64, 339–341.